

Sneeze

Exercise

Laugh

Cough

Walk
then you may suffer from Stress Urinary Incontinence (SUI).
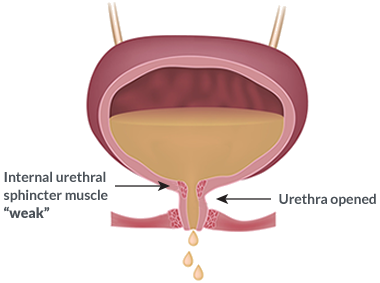
SUI is when urine leaks out during moments of physical activity that increases abdominal pressure, such as coughing, sneezing, laughing, or exercise.
This condition is caused by a weakness of the pelvic floor muscles, which usually tends to happen as we get older, with pregnancy and with certain pelvic floor conditions.

Did you know that stress urinary incontinence is a common condition that affects 1 in 3 women at some point in their lives.1
Dealing with stress urinary incontinence can be frustrating, struggling with embarrassing bladder leaks. But luckily there is a treatment option available to help you regain bladder control.

Bulkamid is a urethral bulking agent that is used to treat stress urinary incontinence. It is a soft, water-based gel that can be used to restore the natural closing of the urethra.
The majority of women with SUI choose Bulkamid before other treatment options.2
Real Stories. Real Results.

Suffering with stress urinary incontinence was embarrassing and terrifying. Now, I am ecstatic, and I love my "new urethra" thanks to Bulkamid! — Nicole M. | Bulkamid Patient —
Results and experiences may vary and are unique to each patient. No promise or guarantee is made about specific results or experiences.
Ready to say goodbye to bladder leaks? Take the first step!
Ask your doctor today if Bulkamid is right for you or get in touch with our team here to get connected with a Bulkamid provider.
References:
IMPORTANT SAFETY INFORMATION:
Indications: The Bulkamid Urethral Bulking System is indicated for urethral injection for the treatment of stress urinary incontinence (SUI) due to intrinsic sphincter deficiency (ISD) in adult women who have SUI or stress predominant mixed incontinence.
Contraindications: Bulkamid Urethral Bulking System must not be used in patients suffering from acute urinary tract infection.
Warnings: Do not inject Bulkamid Hydrogel intravascularly. Injection of Bulkamid Hydrogel into blood vessels may cause vascular occlusion leading to a possible embolism. Discontinue injection of Bulkamid Hydrogel if the superficial capillaries of the mucosa start to fade in order to avoid ischemia. Prior assessment of the tissue is recommended before introducing the Bulkamid Rotatable Sheath into the urethra. Do not force the Bulkamid Rotatable Sheath into the urethra or inject Bulkamid Hydrogel if the urethral tissue is damaged. The Bulkamid Urethral Bulking System should not be used in patients with urethral or bladder neck strictures until the strictures have been corrected. Use of the Bulkamid Urethral Bulking System in patients with strictures may cause injury and/or urethral obstruction. Over-correction using Bulkamid Hydrogel may lead to obstruction. Patients receiving treatment affecting blood coagulation have an increased risk of hematoma or urethral bleeding, as with any invasive procedure. Do not use Bulkamid Hydrogel in male patients.
Precautions: The Bulkamid Urethral Bulking System is only to be administered by a qualified physician, e.g. gynecologist, urologist, or urogynecologist. Safety and effectiveness of Bulkamid have not been established in patients with a fragile urethral mucosal lining, with urethral hypermobility with a straining angle >30º from horizontal bladder neck, predominant urge incontinence, detrusor overactivity, known polyuria (≥ 3L/24h), unevaluated hematuria, prolapse stage greater than Stage II using the ICS Pelvic Organ Prolapse Quantification (POPQ) exam, BMI >35 kg/m2, neurogenic bladder, less than 18 years of age, have active Herpes Genitalis, or for re-injection of Bulkamid Hydrogel less than 4 weeks after initial injection. The effect of Bulkamid has not been evaluated in women during pregnancy, delivery or lactation. The effect of Bulkamid on subsequent pregnancy and delivery, and the impact of subsequent pregnancy on the effect of Bulkamid, is unknown. Therefore, the risks and benefits of the device in women of childbearing potential should be carefully assessed.
Adverse Events: Adverse events may include: pain at the implant site, acute retention, urinary tract infection, hematuria, de novo urge incontinence, dysuria, urinary urgency, vaginal infection/irritation/Lichen Sclerosus, and worsening urinary incontinence.
Caution: U.S. Federal law restricts this device to sale and use by, or on the order of, a physician.
For a complete listing of indications, contraindications, warnings and precautions, go to www.bulkamid.com/isi

© 2024 Axonics, Inc. All rights reserved.
26 Technology Drive, Irvine, CA 92618
Toll-Free: 1-877-9-AXONICS